ECT coordinates funding for two pan-European, non-commercial, investigator-driven clinical trials targeting rare diseases in the pediatric population in compliance with Good Clinical Practice and current national and European legislation and regulations:
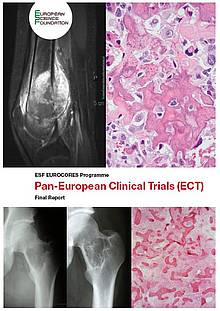
EURAMOS (European and American Osteosarcoma Study Group), a randomised trial designed to optimise treatment strategies for patients with resectable osteosarcoma based on histological response to pre-operative chemotherapy. Osteosarcoma is the most common bone cancer in children, adolescents and young adults. EURAMOS involves over 300 clinical centres in 13 European countries, the USA, Australia, New Zealand, Bahamas, Puerto Rico, Guatemala and Canada, and has already recruited 2 260 patients including over 1 100 in Europe as of October 2009.
More information is available here.PROFIDYS, a randomised placebo-controlled trial designed to assess the safety, tolerability and efficacy of an oral bisphosphonate in the reduction of bone pain and osteolytic lesions in patients with fibrous dysplasia of the bone, a rare congenital bone disease characterised by replacement of normal bone by fibrous tissue. PROFIDYS involves 5 clinical centres in 3 European countries (France, Belgium and the Netherlands). As this disease is so rare, 110 patients are being recruited for this trial, 46 of which were randomised as of 1 January 2010.
Success Story
Upon request of the European Commission, a “Success Story” was written for the EUROCORES Scheme Final Activity Report (Sixth Framework Programme, contract No ERAS-CT-2003-980409, period covered: 01/10/2003 to 31/03/2009)